 Periods are the horizontal rows of the periodic table. #S PERIODIC TABLE PLUS#For d-block elements, the group numbers is (n-1) the number of electrons in the subshell plus the number of electrons in the valence shell. for p-block elements, the group number is 10 plus the number of valence electrons. For s-block elements, the group number is the number of valence electrons. Moving down a group adds a new electron subshell. Groups are the vertical columns on the periodic table.Blocks indicate which electron sublevel is being filled.Blocks vs Groups and Periodsīlocks, groups, and periods are three broad ways of classifying elements based on their position on the periodic table and common properties. As these elements have not yet been synthesized, their properties have yet to be determined. The g-block elements will be elements beyond oganesson (element 118). Many of the f-block elements (the actinides) are radioactive.These elements form colored complexes and salts, but they tend to be paler than those formed by d-block elements.Most f-block elements have high melting points.F-block elements display variable oxidation states.They are the two rows of elements found below the main body of the periodic table. The f-block elements or inner transition metals are the lanthanides and actinides. D-block elements tend to be good catalysts.Many of these elements form colored complexes and salts.D-block elements tend to have high melting points and boiling points.These elements are all metals, usually with two or more oxidation states.This is why they are called “transition” metals. 
D-block elements display properties between those of the highly reactive electropositive s-block elements and the more electronegative p-block elements. 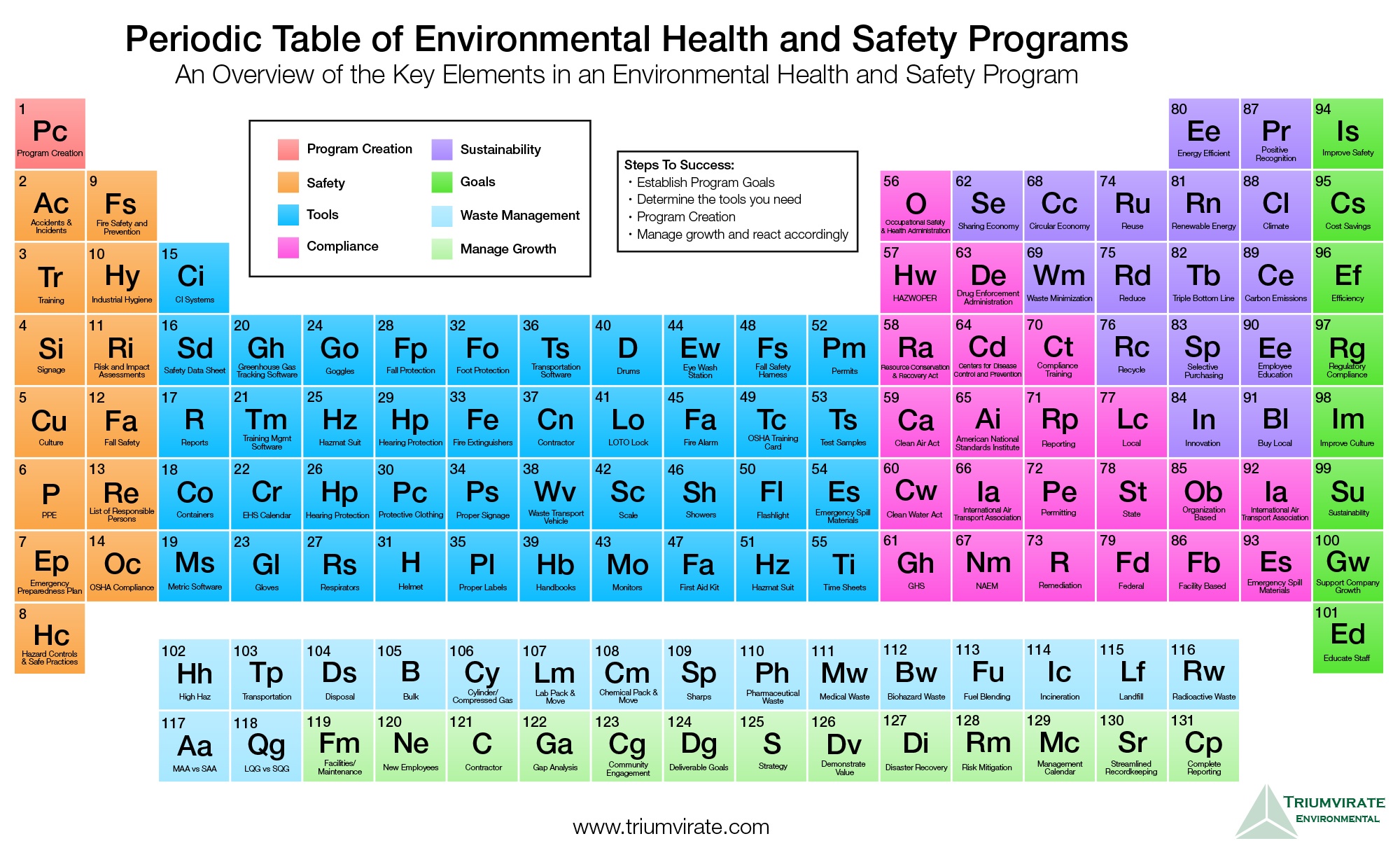
The halogens form ionic compounds with s-block elements.ĭ-block elements are the transition metals (groups 3-12).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
